ODYSSEY VSEPR Theory Hack 1.0.5 + Redeem Codes
Developer: Wavefunction, Inc
Category: Education
Price: $3.99 (Download for free)
Version: 1.0.5
ID: com.wavefun.OdyVSEPR
Screenshots
Description
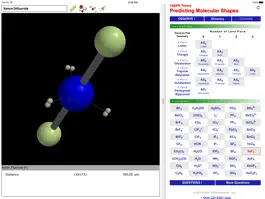
Why are some triatomic molecules linear (carbon dioxide, hydrogen cyanide) and others are bent (water, sulfur dioxide)? And why is the bond angle in water (~105°) smaller than that in sulfur dioxide (~119°)? Valence Shell Electron Pair Repulsion (VSEPR) theory provides surprisingly simple explanations in terms of the electron pairs that surround the central atom of a given molecule. VSEPR theory is described in detail in every introductory chemistry book.
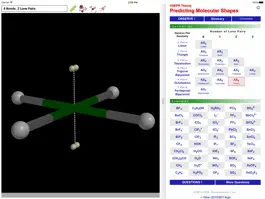
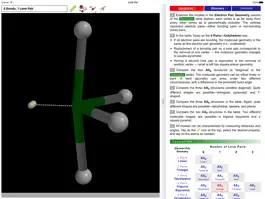
The ODYSSEY VSEPR Theory app provides three-dimensional models of 50 small molecules and ions as well as a set of generic models that illustrate the conceivable geometries of the theory. Each model can be moved and manipulated with simple touch gestures. Bond lengths and bond angles can be queried.
Users can pick from multiple model styles (such as Ball and Spoke or Space Filling), request a schematic display of lone pair positions, and show the molecular dipole arrow. A glossary, comments section, and a set of multiple-choice questions (with randomized options) are also available.
Most learners of chemistry are visually oriented. Complementing the presentation found in any introductory textbook, ODYSSEY VSEPR Theory enables exploration of one of the most fundamental concepts in general chemistry—molecular shape.
The ODYSSEY VSEPR Theory app provides three-dimensional models of 50 small molecules and ions as well as a set of generic models that illustrate the conceivable geometries of the theory. Each model can be moved and manipulated with simple touch gestures. Bond lengths and bond angles can be queried.
Users can pick from multiple model styles (such as Ball and Spoke or Space Filling), request a schematic display of lone pair positions, and show the molecular dipole arrow. A glossary, comments section, and a set of multiple-choice questions (with randomized options) are also available.
Most learners of chemistry are visually oriented. Complementing the presentation found in any introductory textbook, ODYSSEY VSEPR Theory enables exploration of one of the most fundamental concepts in general chemistry—molecular shape.
Version history
1.0.5
2022-07-29
iOS 15 fixes- minor content updates
1.0.4
2018-12-07
Compatibility fix for iOS 10 and later.
1.0.3
2016-01-21
- iPad Pro compatibility
- Quarter point penalty for incorrect answers removed
- Quarter point penalty for incorrect answers removed
1.0.2
2014-12-03
- Compatibility fixes for iOS 8.1
- Touch interface atom picking improved
- Touch interface atom picking improved
1.0.1
2014-10-10
- Fix for retina displays under iOS 8
- Improved presentation
- Improved presentation
1.0.0
2014-05-06
Ways to hack ODYSSEY VSEPR Theory
- Redeem codes (Get the Redeem codes)
Download hacked APK
Download ODYSSEY VSEPR Theory MOD APK
Request a Hack
Ratings
5 out of 5
1 Ratings